About the Study
The EQUAL study is evaluating a new blood test to detect a specific type of lung cancer in individuals at risk for the disease and who would otherwise be unable to undergo routine lung cancer screening. We are also working to understand perceptions of lung cancer and the concerns, perspectives, and experiences that influence participants’ choices in using our blood test for lung cancer screening, in order to optimize the blood test to improve early detection options for lung cancer.

Who is eligible to join the EQUAL Study?
Group #1: Non-tobacco using (currently), 50–80-year-olds, who self-identify as East Asian (including Southeast Asian) or Latinx
- East Asian includes those who self-identify as Chinese, Japanese, Korean, Taiwanese, Malaysian
- Southeast Asian includes those who self-identify as Cambodian, Thai, Vietnamese, Filipino
- Latinx includes those who self-identify as a person of Central or South American and the Caribbean including, Cuban, Puerto Rican, and Dominican culture or origin, excluding individuals originating from Spain.
Group 2: Non-tobacco using (currently), 40-49-year-olds, who self-identify as East Asian (including Southeast Asian) or Latinx, if any of the following criteria are met:
- Family history of any EGFR mutant LC
- Personal history of remote cancer that is not lung cancer or other thoracic malignancies, including thymoma, thymic carcinoma, or sarcoma, as long as it was resolved over 5 years ago
- History of tuberculosis, asthma requiring daily inhaled corticosteroids or chronic bronchitis
- Symptoms of lung cancer for the past month, including hemoptysis, unexplained weight loss, voice hoarseness, cough or worsening cough, dyspnea or worsening dyspnea
How can I join the EQUAL Study?
You may also contact the study team at EQUALstudy@dfci.harvard.edu
Why should I join?
Taking part in this research study may not directly benefit you. However, the information gained from your participation will likely help researchers learn information on the EQUAL study blood test's ability to screen for EGFR lung cancer, as well as about participants' perceptions of lung cancer screening and their concerns, perspectives, and experiences related to undergoing lung cancer screening with EQUAL's blood test.
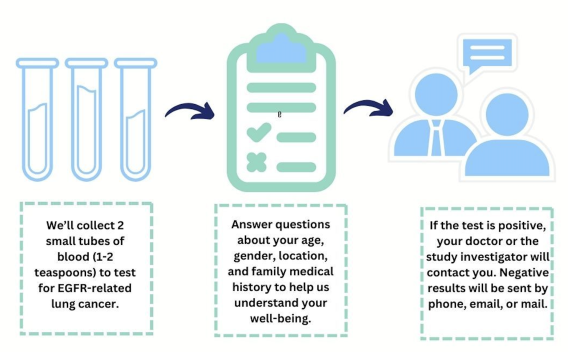
If you do test positive with our blood test, we may be able to treat you early, before the disease spreads. We will also connect you to a healthcare provider for lung cancer management at your preferred institution.
What is involved?
After confirming eligibility for the EQUAL study, two tubes (approximately 1-2 tablespoons) of blood will be collected from you at a study-site hospital or at your home through a mobile phlebotomist.
We will also ask you to complete a short questionnaire about your family and personal medical history, your family medical history, social factors like housing, education and income and other questions related to your overall health.
EQUAL study participants will also have the option to participate in an optional survey and/or a focus group to share their perceptions and concerns about using a blood-based lung cancer screening test.
Additionally, participants can take part in a separate blood banking study being conducted at Dana-Farber Cancer Institute, called the InAdvance study.
Our Mission
To bring state-of-the-art early detection and precision interventions to people at higher risk of developing cancer and to foster collaboration between researchers
Our Partners



